Steel is one of the most resilient and versatile materials in building and construction. However, for all its strength, steel will eventually rust if not treated properly.
Steel corrosion can be defined as deterioration due to electrochemical reactions between the metal and its environment. There are different types of corrosion, which are contingent on different environmental factors. One of these is galvanic corrosion.

Galvanic corrosion can wreak havoc on steel sections, and it looks terrible. Here we take a look at why it occurs and how you can prevent it.
What Is Galvanic Corrosion?
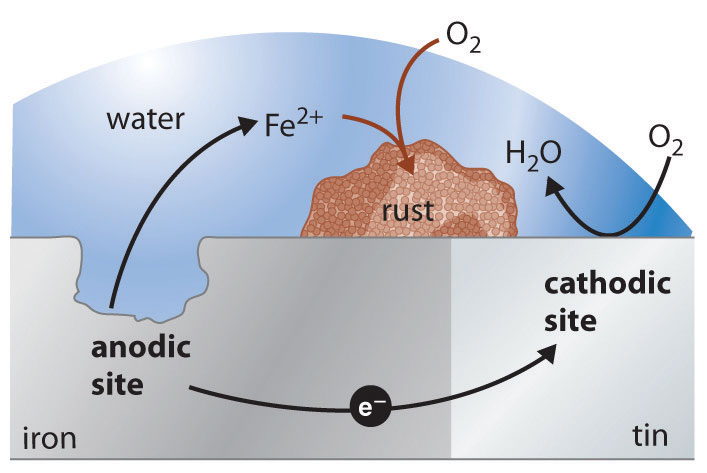
Galvanic corrosion is also known as dissimilar metal corrosion, and as the latter suggests, it occurs when steel comes into contact with another metal in a corrosive electrolyte.
What Causes Galvanic Corrosion?
Galvanic corrosion occurs when multiple dissimilar metal types come into contact in the presence of of conductive substance; one metal becomes anodic – meaning that it will corrode faster – while part of the remaining metal becomes cathodic – meaning that it will corrode slower, or not at all. Usually it is the more ‘active’ metal (more likely to become active in a given electrolyte) that becomes an anode (a positively charged electrode).
For galvanic corrosion to occur, three things are required:
• Electrochemically dissimilar materials
• Contact between the two metals
• The two metals exposed to the same conduction solution (such as water)
It is impossible for corrosion to occur unless all three elements are present, and thus, protecting against galvanic corrosion is simple – eliminate an element. This is best achieved by ensuring the two metals do not touch by establishing a barrier between them.
The Galvanic Series List
The galvanic series lists arrange common metals in order from the most anodic to most cathodic. The further apart the metals are on the list, the faster the corrosion rate will be. There are different lists for different conductive solutions, such as saltwater and freshwater, which will impact the speed of corrosion also.

Protecting Against Steel Corrosion With Steel Coating
Surface treatment is the most effective way to protect against corrosion. Here are some of the most common you will see steel fabricators use:
Hot dipping – This galvanisation method can be used on steel of all shapes and sizes and involves immersing the steel in a bath of molten zinc at temperatures up to 450°C. Steel galvanised in this way is highly protected against corrosion as well as extreme weather conditions and this is a particularly popular approach for pipe related applications.
Zinc phosphate priming – Steel surfaces can be painted with specially formulated primers in order to improve corrosion resistance as well as boost their visual appeal, with one of the most popular applications being zinc phosphate priming.
Chemical coating – This brilliant technique uses electrostatic or compressed air to apply a specially formulated powdered material to the steel surface, following which it is melted to form a smooth protective film. Steel treated in this way is not only protected against corrosion and UV damage, it is also highly resistant to peeling, scratching and cracking.
Zinc spray metallising – While it doesn’t provide quite the same protection as hot dip galvanising, this technique is highly effective against corrosion and is popular for its smooth finish. Because this is a ‘cold process’ there is no risk of distorting the metal, making it ideal for use on ornate metal components such as railings and fences.
Need Help With A Steel Project?
If you have a project in need of steel, whether it’s supply, fabrication, drafting, or installation, talk to the experts at Steel Fabrication Services. Our dedicated team of professionals will ensure that your project goes according to plan right down to the smallest detail. Give us a call today!
While you’re here, learn more about fighting corrosion below:
